Enthalpy Of Vaporization Of Methanol
Methanol, CHthree-OH, is a colorless, adequately volatile liquid with a faintly sweet pungent scent, similar, but somewhat milder and sweeter than ethanol.
Methanol is toxic, and may cause blindness. The vapors are slightly heavier than air and may explode if ignited.
Methanol is used to brand chemicals, to removewater from automotive and aviation fuels, as a solvent for paints and plastics, and every bit an ingredient in a wide variety of products.
The phase diagram of methanol is shown beneath the table.
Chemical, physical and thermal properties of ethanol:
Values are given for liquid at 25oC /77oF / 298 Thousand and ane bara, if not other stage, temperature or pressure given.
For full tabular array with Regal Units - rotate the screen!
| Property | Value | Unit | Value | Unit | Value | Unit | Value | Unit | |||
| Acerbity (pKa1) | xv.3 | ||||||||||
| Autoignition temperature | 737 | K | 464 | °C | 867 | °F | |||||
| Boiling Point | 337.85 | K | 64.7 | °C | 148.5 | °F | |||||
| Critical density | eight.7852 | mol/dm3 | 281 | kg/miii | 0.546 | slug/ftiii | 17.6 | lb/ft3 | |||
| Critical Pressure | 8.22 | MPa=MN/mtwo | 82.2 | bar | 81.1 | atm | 1192 | psi=lbf/inii | |||
| Critical temperature | 513.4 | Chiliad | 240.2 | °C | 464.4 | °F | |||||
| Critical Book | 114 | cm3/mol | 0.003552456 | m3/kg | ane.831 | ft3/slug | 0.05690 | ftthree/lb | |||
| Density (gas) at 0.17 bar | half dozen.87 | mol/m3 | 0.220 | kg/m3 | 0.00043 | slug/ftiii | 0.0137 | lb/ft3 | |||
| Density (liquid) | 24540 | mol/m3 | 786.3 | kg/thou3 | ane.526 | slug/ftiii | 49.09 | lb/ft3 | |||
| Combustible, gas and liquid | yes | ||||||||||
| Flash point | 284 | One thousand | 11 | °C | 52 | °F | |||||
| Gas abiding, individual - R | 259.5 | J/kg Grand | 0.07208 | Wh/(kg K) | 1552 | [ft lbf/slug °R] | 48.23 | [ft lbf/lb °R] | |||
| Gibbs complimentary energy of formation (gas) | -163 | kJ/mol | -5087 | kJ/kg | -2187 | Btu/lb | |||||
| Heat (enthalpy) of combustion (gas) | 726.one | kJ/mol | 22661 | kJ/kg | 9.vii | Btu/lb | |||||
| Heat (enthalpy) of combustion (liquid) | -727 | kJ/mol | -22689 | kJ/kg | -9.8 | Btu/lb | |||||
| Heat (enthalpy) of germination (gas) | -205 | kJ/mol | -6398 | kJ/kg | -2751 | Btu/lb | |||||
| Heat (enthalpy) of formation (liquid) | -239 | kJ/mol | -7459 | kJ/kg | -3207 | Btu/lb | |||||
| Heat (enthalpy) of fusion at -143°F/-97°C | 3.18 | kJ/mol | 99 | kJ/kg | 42.67 | Btu/lb | |||||
| Heat (enthalpy) of evaporation | 37.34 | kJ/mol | 1165 | kJ/kg | 501.01 | Btu/lb | |||||
| Specific heat chapters, Cp (gas) at 0.17 bar | 116 | J/mol 1000 | 3.62 | kJ/kg K | 0.865 | Btu/lb°F or cal/thousand G | |||||
| Specific oestrus chapters, Cp (liquid) | 81.two | J/mol One thousand | ii.53 | kJ/kg K | 0.605 | Btu/lb°F or cal/g 1000 | |||||
| Specific oestrus capacity, Cp (solid) at -148°F/-100°C | 105 | J/mol Grand | 3.28 | kJ/kg K | 0.783 | Btu/lb°F or cal/g Yard | |||||
| Specific heat capacity, Cv (gas) at 0.17 bar | 94.5 | J/mol K | 2.95 | kJ/kg K | 0.704 | Btu/lb°F or cal/g K | |||||
| Specific rut chapters, Cv (liquid) | 67.ix | J/mol Grand | 2.12 | kJ/kg Thousand | 0.506 | Btu/lb°F or cal/one thousand G | |||||
| Ionization potential | 10.84 | eV | |||||||||
| log KOW (Octanol/Water Partition Coefficient) | -0.77 | ||||||||||
| Melting bespeak | 175.55 | K | -97.6 | °C | -143.seven | °F | |||||
| Molecular Weight | 32.042 | g/mol | 0.07064 | lb/mol | |||||||
| Solubility in water | 1000 | mg/ml | (Miscible in h2o) | ||||||||
| Audio velocity in gas, at 0.17 bar | 297 | yard/s | 974 | ft/s | 665 | mi/h | |||||
| Sound velocity in liquid | 1100 | m/s | 3608 | ft/s | 2464 | mi/h | |||||
| Specific Gravity (gas) (relativ to air) | 1.xi | ||||||||||
| Specific Gravity (liquid) (relativ to water) | 0.79 | ||||||||||
| Specific Heat Ratio (gas) - cp/cv | i.23 | ||||||||||
| Specific Heat Ratio (liquid) - cp/cv | i.20 | ||||||||||
| Specific Book (gas) 0.17 bar | 0.146 | m3/mol | four.55 | m3/kg | 2342.63 | ft3/slug | 72.81 | ftthree/lb | |||
| Specific Volume (liquid) | 0.0000408 | chiliadiii/mol | 0.00127 | mthree/kg | 0.655 | ft3/slug | 0.0204 | ft3/lb | |||
| Standard molar entropy, Southward° (gas) | 240 | J/mol K | 7.49 | kJ/kg K | 1.79 | Btu/lb °F | |||||
| Standard molar entropy, S° (liquid) | 127 | J/mol K | iii.96 | kJ/kg Grand | 0.95 | Btu/lb °F | |||||
| Surface tension | 22.07 | dynes/cm | 0.02207 | N/thousand | |||||||
| Thermal Conductivity | 0.202 | W/chiliad°C | 0.116713 | Btu/hr ft °F | |||||||
| Triple betoken force per unit area | one.86*10-7 | MPa=MN/m2 | 1.86*ten-half dozen | bar | ane.84*10-6 | atm | 2.70*10-5 | psi=lbf/inii | |||
| Triple bespeak temperature | 175.50 | K | -97.7 | °C | -143.77 | °F | |||||
| Vapor (saturation) pressure | 0.0169 | MPa=MN/10002 | 127.0 | mm Hg | 0.1671 | atm | 2.46 | psi=lbf/inii | |||
| Viscosity, dynamic (absolute) | 0.544 | cP | 365.55 | [lbm /ft southward*10-6] | 11.36 | [lbf due south/fttwo *10-six] | |||||
| Viscosity, kinematic | 0.692 | cSt | 7.iv | [fttwo/s*10-six] | |||||||
Back to top
Follow the links beneath to go values for the listed properties of methanol at varying pressure level and temperature:
- Density and specific weight
- Dynamic and kinematic viscosity
- Specific heat (heat capacity)
See as well more than almost atmospheric pressure, and STP - Standard Temperature and Pressure & NTP - Normal Temperature and Pressure,
every bit well as Thermophysical properties of: Acetone, Acetylene, Air, Ammonia, Argon, Benzene, Butane, Carbon dioxide, Carbon monoxide, Ethane, Ethanol, Ethylene, Helium, Hydrogen, Hydrogen sulfide, Methane, Nitrogen, Oxygen, Pentane, Propane, Toluene, Water and Heavy h2o, D2O.
Methanol is a liquid at standard atmospheric condition. However, at low temperature and/or very loftier pressures information technology becomes a solid.
The phase diagram for methanol shows the stage behavior with changes in temperature and pressure. The curve between the disquisitional point and the triple point shows the methanol boiling betoken with changes in pressure. It also shows the saturation pressure with changes in temperature.
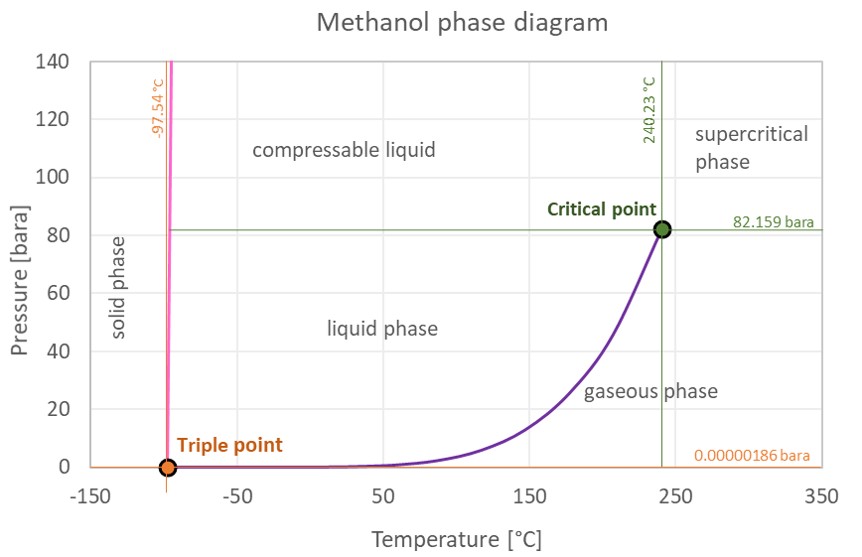
At the critical betoken at that place is no change of country when pressure is increased or if estrus is added.
The triple point of a substance is the temperature and pressure at which the 3 phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.
Enthalpy Of Vaporization Of Methanol,
Source: https://www.engineeringtoolbox.com/methanol-methyl-alcohol-properties-CH3OH-d_2031.html
Posted by: vanhorneworidence.blogspot.com


0 Response to "Enthalpy Of Vaporization Of Methanol"
Post a Comment